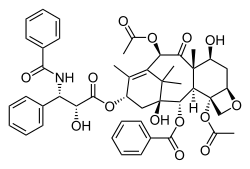
Paclitaxel (Taxol) is a potent anti-neoplastic and anti-mitotic taxane compound used in cancer chemotherapy. Paclitaxel binds to the N-terminus of β-tubulin and and stabilizes microtubules arresting the cell cycle at the G2/M phase. The microtubule damage induces apoptosis through a JNK-dependent pathway followed by a JNK-independent pathway, perhaps related to the activation of protein kinase A (PKA) or of Raf-1 kinase, resulting in phosphorylation of Bcl-2. Paclitaxel stabilizes microtubules and as a result, interferes with the normal breakdown of microtubules during cell division. The ability of paclitaxel to inhibit spindle function is generally attributed to its suppression of microtubule dynamics, but recent studies have demonstrated that suppression of dynamics occurs at concentrations lower than those needed to block mitosis. At the higher therapeutic concentrations, paclitaxel appears to suppress microtubule detachment from centrosomes, a process normally activated during mitosis. The binding site for paclitaxel has been identified on the beta-tubulin subunit. In vitro and in vivo studies have demonstrated that paclitaxel is extensively metabolised by the liver to 3 primary metabolites.

J Virol. 2022 Jul 27;96(14):e0212721.
PRRSV infection induces gasdermin d-driven pyroptosis of porcine alveolar macrophages through NLRP3 inflammasome activation
Paclitaxel purchased from AbMole

Transl Lung Cancer Res. 2021 Feb;10(2):1007-1019.
Triptolide inhibits epithelial-mesenchymal transition phenotype through the p70S6k/GSK3/β-catenin signaling pathway in taxol-resistant human lung adenocarcinoma
Paclitaxel purchased from AbMole

Drug Deliv. 2021 Dec;28(1):2256-2267.
GLUT1 targeting and hypoxia-activating polymer-drug conjugate-based micelle for tumor chemo-thermal therapy
Paclitaxel purchased from AbMole

Chem Pharm Bull (Tokyo). 2021;69(4):325-332.
MiR-509-3p Induces Apoptosis and Affects the Chemosensitivity of Cervical Cancer Cells by Targeting the RAC1/PAK1/LIMK1/Cofilin Pathway
Paclitaxel purchased from AbMole

Oncogene. 2020 Sep;39(37):6024-6040.
CRMP2 is a therapeutic target that suppresses the aggressiveness of breast cancer cells by stabilizing RECK
Paclitaxel purchased from AbMole
Cell Cycle. 2018;17(10):1235-1244.
Apatinib, a novel tyrosine kinase inhibitor, suppresses tumor growth in cervical cancer and synergizes with Paclitaxel.
Paclitaxel purchased from AbMole
Cancer Biol Ther. 2018 Feb 5.
Buformin suppresses proliferation and invasion via AMPK/S6 pathway in cervical cancer and synergizes with paclitaxel
Paclitaxel purchased from AbMole
| Cell Experiment | |
|---|---|
| Cell lines | human neonatal dermal microvascular ECs (HMVECs), human umbilical vein ECs (HUVECs), human umbilical artery ECs (HUAVECs), normal human astrocytes (NHAs), normal human dermal fibroblasts (NHDFs), normal human epidermal keratinocytes (NHEKs |
| Preparation method | Cells including human neonatal dermal microvascular ECs (HMVECs), human umbilical vein ECs (HUVECs), human umbilical artery ECs (HUAVECs), normal human astrocytes (NHAs), normal human dermal fibroblasts (NHDFs), normal human epidermal keratinocytes (NHEKs), human mammary epithelial cells (HMEpCs), human prostate epithelial cells (PrEpCs) and human umbilical artery smooth muscle cells (UASMCs) are cultured. Cell proliferations are performed in 96-well plates using cells between passages 6 and 12. Cells are seeded at 3000–5000 cells/well and allowed to attach for 4 hours. Paclitaxel, diluted in culture medium, is added in quadruplicate wells and the cells ae incubated for 3 days before MTS reagents are added to quantitate the live cells in each well. |
| Concentrations | 0.1-100 pM |
| Incubation time | 72 h |
| Animal Experiment | |
|---|---|
| Animal models | nude athymic mice with BC-V and BC-ER tumors |
| Formulation | Dissolved in absolute ethanol with an equal volume of cremophor and diluted 1:4 with sterile physiological saline before use |
| Dosages | 20 mg/kg |
| Administration | i.v. |
| Molecular Weight | 853.91 |
| Formula | C47H51NO14 |
| CAS Number | 33069-62-4 |
| Solubility (25°C) | DMSO 50 mg/mL |
| Storage | -20°C, protect from light, sealed |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
[3] Sonnichsen DS, et al. Clin Pharmacokinet. Clinical pharmacokinetics of paclitaxel.
| Related Microtubule Products |
|---|
| P34cdc2 Kinase Fragment
P34cdc2 Kinase Fragment is associated with the completion of DNA replication in yeast mitosis. |
| Cemadotin
Cemadotin (LU103793) is an analogue of Dolastatin 15 which is naturally occurring cytotoxic peptides. |
| VLS-1488
VLS-1488 is an orally active KIF18A inhibitor that inhibits KIF18A microtubule-dependent ATPase activity with an IC50 value of 0.016 μM. |
| ENMD-1198
ENMD-1198 (IRC-110160), an orally active microtubule destabilizing agent, is a 2-methoxyestradiol analogue with antiproliferative and antiangiogenic activity. |
| Tubulin/JAK2-IN-1
Tubulin/JAK2-IN-1 is a dual inhibitor of Janus kinase 2 (JAK2) and microtubule. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
