All AbMole products are for research use only, cannot be used for human consumption.
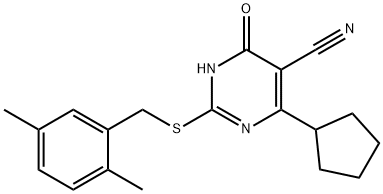
HJC0197 is a potent antagonist of Epac1 (the exchange protein directly activated by cAMP 1) and Epac2 (IC50=5.9 μM for Epac2). HJC0197 selectively blocked camp-induced Epac activation. HJC0197 inhibited epAC1-mediated RAP1-GDP exchange activity in the presence of isoconcentration of cAMP.
| Molecular Weight | 339.45 |
| Formula | C19H21N3OS |
| CAS Number | 1383539-73-8 |
| Solubility (25°C) | DMSO ≥ 40 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
[5] Holger Rehmann. Sci Rep. Epac-inhibitors: facts and artefacts
| Related Ras Products |
|---|
| AMG410
AMG410 is a non-covalent and selective pan-KRAS inhibitor with IC50 values of 1-4 nM for KRAS G12D, KRAS G12V, and KRAS G13D. AMG410 is a dual GTP(on)- and GDP(off)-state inhibitor (Kd(GDP-state) of 1 nM; Kd(GTP-state) of 22 nM). AMG410 blocks KRAS signaling in a cycling state-independent manner and also blocks proliferation in wildtype KRAS-amplified tumor cells. |
| HRS-4642
HRS-4642 is a selective KRAS(G12D) inhibitor, with Kd value of 0.083 nM. HRS-4642 has anti-cancer activity. HRS-4642 exhibits significant in vivo potency. |
| AZD0022
AZD0022 is a selective and orally active KRASG12D inhibitor. |
| HH0043
HH0043 is an orally active and potent SOS1 inhibitor, with IC50 value of 5.8 nM. |
| BI-2493
BI-2493 is a structural analogue of BI-2865 and a highly selective and orally active pan-KRAS inhibitor. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
