All AbMole products are for research use only, cannot be used for human consumption.
For this product's availability, delivery time and price, please email [email protected] directly or click the "Inquiry Now" button below.
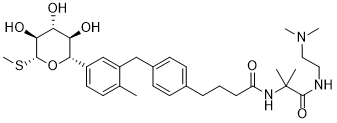
LX2761 is a locally acting SGLT1 and SGLT2 inhibitor in vitro with IC50 values of 2.2 nM and 2.7 nM for hSGLT1 and hSGLT2, respectively. LX2761 is a very potent, chemically stable, and luminally restricted molecule that achieved proof of concept in OGTT at very low doses. LX2761 was designed to remain in the intestine after oral delivery to inhibit SGLT1 locally without affecting the SGLT1/2 mechanism in the kidney.
| Molecular Weight | 601.8 |
| Formula | C32H47N3O6S |
| CAS Number | 1610954-97-6 |
| Storage | -20°C, dry, protect from light, sealed |
| Related SGLT Products |
|---|
| Dapagliflozin
Dapagliflozin (BMS-512148) is a selective, orally active inhibitor of the renal sodium-glucose co-transporter type 2 (SGLT2). |
| Canagliflozin
Canagliflozin is a potent, selective sodium glucose co-transporter 2 inhibitor. |
| Empagliflozin
Empagliflozin (BI-10773) is a potent and selective SGLT-2 inhibitor with IC50 of 3.1 nM, exhibits >300-fold selectivity over SGLT-1, 4, 5 and 6. |
| Phloridzin
Phloridzin is a non-selective SGLT inhibitor with Kis of 300 and 39 nM for hSGLT1 and hSGLT2, respectively. Phlorizin is also a Na+/K+-ATPase inhibitor. |
| Phloretin
Phloretin (NSC 407292; RJC 02792) is a specific, competitive and orally active inhibitor of sodium/glucose cotransporters in the intestine (SGLT1) and kidney (SGLT2). |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.
Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2026 AbMole BioScience. All Rights Reserved.