All AbMole products are for research use only, cannot be used for human consumption.
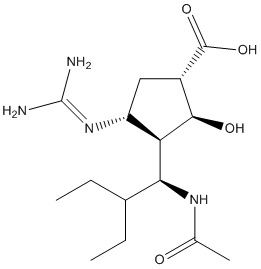
Peramivir is a transition-state analogue and a potent, specific influenza viral neuraminidase inhibitor with an IC50 of median 0.09 nM. The IC50 for peramivir is markedly lower than that for either zanamivir or oseltamivir. Peramivir increases survival in preclinical influenza models, including mouse and ferret models of highly pathogenic avian influenza H5N1. One day of treatment with peramivir is active, and multiple days of treatment rescues most or all mice with H5N1. Similar data have been obtained in ongoing murine experiments with seasonal influenza A (H1N1)/H274Y. After parenteral administration, peramivir reaches very high plasma concentrations. Peramivir is not metabolized, and is cleared by the means of renal filtration. It has a long half-life, especially in comparison with other neuraminidase inhibitors. Due to the failure of the metabolism of the antivirus compound peramivir, as well as its wide distribution and the excretion in the unchanged form in urine, dosing regimens can be adapted easily for patients with renal impairment and for pediatric populations. Peramivir is originally developed by BioCryst Pharmaceuticals. And peramivir is designed to aim to an antiviral agent. Peramivir has been performed phase III clinical trials for the treatment of influenza.
Another CAS# 229614-55-5

EBioMedicine. 2017 Aug;22:133-142.
C-Reactive Protein Mediating Immunopathological Lesions: A Potential Treatment Option for Severe Influenza A Diseases
Peramivir purchased from AbMole
| Molecular Weight | 328.41 |
| Formula | C15H28N4O4 |
| CAS Number | 330600-85-6 |
| Solubility (25°C) | 10 mM in DMSO |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
[1] No authors listed. Peramivir
[2] No authors listed. Aust Prescr. Peramivir for influenza
[3] Lesley J Scott. Peramivir: A Review in Uncomplicated Influenza
[4] No authors listed. Peramivir
[5] Milena M McLaughlin, et al. Peramivir: an intravenous neuraminidase inhibitor
| Related Anti-infection Products |
|---|
| Strictinin
Strictinin is a phenolic compound which has potential antiviral, antibacterial and laxative activities. Strictinin occurs by accelerating intestinal transit rather than enhancing gastric emptying, increasing food intake, or inducing diarrhea in rats. |
| Torcitabine
Torcitabine (2'-Deoxy-L-cytidine) is an antiviral agent. Torcitabine has the potential for chronic hepatitis B virus infection research. Torcitabine (2'-Deoxy-L-cytidine) shows greater inhibition of first strand than second strand DNA synthesis. |
| ABMA
ABMA is a broad-spectrum inhibitor of intracellular toxins and pathogens. ABMA efficiently protects cells against various toxins and pathogens including viruses, intracellular bacteria and parasite. |
| DDX3-IN-1
DDX3-IN-1 is a DEAD-box polypeptide 3 (DDX3) inhibitor with CC50 values of 50 and 36 μM for HIV and HCV, respectively. DDX3-IN-1 has antiviral activity. |
| K22
K22 is an inhibitor of coronavirus RNA synthesis that specifically targets membrane-associated coronavirus RNA synthesis. K22 effectively blocks replication of multiple coronaviruses by inhibiting the critical step of viral replication complex anchoring to host cell membranes to form double-membrane vesicles (DMVs). K22 exhibits broad-spectrum antiviral activity against diverse coronaviruses. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
