All AbMole products are for research use only, cannot be used for human consumption.
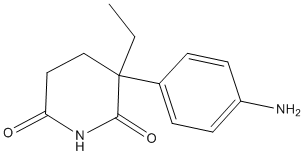
Aminoglutethimide (Cytadren) is an inhibitor of adrenocortical steroid synthesis. It blocks aromatase in the generation of estrogens from androstenedione and testosterone. It also blocks the conversion of cholesterol to pregnenolone by inhibiting the enzyme P450scc and consequently decreases synthesis of all hormonally active steroids. It inhibited bovine adrenal cholesterol side-chain cleavage activity with an IC50 value of 40 μM.
| Cell Experiment | |
|---|---|
| Cell lines | Adrenocortical tumor cell line NCI-h295 |
| Preparation method | Maintaining the NCI-h295 tumor cell line in RPMI 1640 medium which is supplemented with transferrin (0.1 mg/mL), insulin (5 μg/mL), selenium (5.2 μg/mL) and 2% FCS. Incubating the cells for 48 hours with Aminoglutethimide (3, 30, 300 μM). Then examining cells by trypan blue staining for cell viability, counting with a coulter counter. For the assessment of ACTH-R mRNA, cells are harvested, and total RNA is extracted, electrophoresed, blotted and hybridized with a human ACTH-R cDNA probe. |
| Concentrations | Dissolved in 65% ethanol as a stock solution, final concentrations 3, 30, 300 μM. |
| Incubation time | 48 hours |
| Animal Experiment | |
|---|---|
| Animal models | Swiss CD1 male and female mice |
| Formulation | Dissolved in DMSO and diluted in saline. |
| Dosages | 150 mg/kg |
| Administration | Injection |
| Molecular Weight | 232.28 |
| Formula | C13H16N2O2 |
| CAS Number | 125-84-8 |
| Solubility (25°C) | DMSO 10 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
[1] G Cocconi. First generation aromatase inhibitors--aminoglutethimide and testololactone
[2] No authors listed. Lancet. Aminoglutethimide
[3] S W Hughss, et al. Aminoglutethimide: a "side-effect" turned to therapeutic advantage
| Related Products |
|---|
| Poly-L-lysine (MW 150000~300000)
Poly-L-lysine |
| N-Phenylacrylamide
N-Phenylacrylamide |
| Silver diethyldithiocarbamate
Silver diethyldithiocarbamate is used in conjunction with Fe2+ to detect NO in brain, kidney, liver, and other tissues. |
| Methylmalonic acid
Methylmalonic acid can be used as a substrate in esterification reactions by various catalysts. |
| Sarcosine
Sarcosine is a glycine transporter type 1 (GlyT) inhibitor and an N-methyl-D-aspartate (NMDA) receptor co-agonist at the glycine binding site. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.
Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2026 AbMole BioScience. All Rights Reserved.