All AbMole products are for research use only, cannot be used for human consumption.
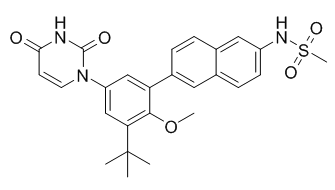
ABT-333 is a nonnucleoside NS5B polymerase inhibitor for the treatment of HCV infection. An interferon-free combination of the protease inhibitor ABT-450 with ritonavir (ABT-450/r), the nonnucleoside polymerase inhibitor ABT-333, and ribavirin showed efficacy against the hepatitis C virus (HCV) in a pilot study involving patients with HCV genotype 1 infection. ABT-333 is currently under clinical trials for the treatment of Hepatitis C.

Nat Commun. 2025 Jan 14;16(1):617.
Oncogenic role of RARG rearrangements in acute myeloid leukemia resembling acute promyelocytic leukemia
Dasabuvir (ABT-333) purchased from AbMole
| Cell Experiment | |
|---|---|
| Cell lines | HCV replicon cells |
| Preparation method | MTT analysis Both replicon-containing cell lines were maintained in Dulbecco’s modified Eagle’s medium (DMEM) containing 100 IU/ml penicillin, 100 g/ml streptomycin, and 200 g/ml G418 (all from Invitrogen, Carlsbad, CA) with 10% (vol/vol) fetal bovine serum (FBS).TheinhibitoryeffectsofdasabuvironHCVRNAreplication were determined in DMEM containing 5% FBS, with or without 40% human plasma (Bioreclamation, Westbury, NY), by measuring the activity of the luciferase reporter gene. The cells were incubated for 3 days in the presence of dasabuvir and subsequently were lysed and processed according to the manufacturer’s instructions (Promega, Madison, WI). The luciferase activity in the cells was measured using a Victor II luminometer (Perkin-Elmer, Waltham, MA); 50% effective concentration (EC50) values were calculated using nonlinear regression curve fitting to a 4-parameter logistic equation with GraphPad Prism 4 software. EC50s and standard deviations (SDs) were calculated from at least 3 experiments conducted in duplicate. The cytotoxicity of dasabuvir was determined with the colorimetric assay using MTT. |
| Concentrations | Dasabuvir inhibited replication of HCV subgenomic replicons in cell culture assays, with EC50 values of 7.7 and 1.8 nM against genotype 1a (H77) and 1b (Con1), respectively. |
| Incubation time | 3 days |
| Animal Experiment | |
|---|---|
| Animal models | none |
| Formulation | |
| Dosages | |
| Administration | |
| Molecular Weight | 493.57 |
| Formula | C26H27N3O5S |
| CAS Number | 1132935-63-7 |
| Solubility (25°C) | DMSO 26 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Related HCV Protease Products |
|---|
| ITMN 4077
ITMN 4077 is a macrocyclic inhibitor against Hepatitis C Virus (HCV) NS3 protease (EC50: 2131 nM). |
| Hepatitis C Virus S5A/5B
Hepatitis C Virus S5A/5B is a synthetic peptide substrate. |
| Ac-D-DGla-LI-Cha-C
Ac-D-DGla-LI-Cha-C is a potent HCV protease inhibitor peptide. |
| Ac-EEVVAC-pNA
Ac-EEVVAC-pNA is a chromogenic substrate for a continuous spectrophotometric assay of HCV NS3 protease. |
| Hepatitis Virus C NS3 Protease Inhibitor 2
Hepatitis Virus C NS3 Protease Inhibitor 2 is a product-based peptide inhibitor of hepatitis C virus (HCV) NS3 protease, with a Ki of 41 nM. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
