All AbMole products are for research use only, cannot be used for human consumption.
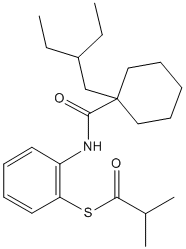
Dalcetrapib (JTT-705) is a cholesteryl ester transfer protein (CETP) inhibitor for rhCETP with an IC50 of 0.2 μM. Dalcetrapib modulates CETP activity. Dalcetrapib induces a conformational change in CETP, when added to human plasma. CETP-induced pre-β-HDL formation in human plasma is unchanged by Dalcetrapib ≤3 µM and increased at 10 µM. Dalcetrapib statistically and significantly increases pre-β-HDL formation.Dalcetrapib achieves 50% inhibition of CETP activity in human plasma at a concentration of 9 μM. Dalcetrapib inhibits the CETP activity of media in HepG2 in a dose-dependent manner. Treatment with Dalcetrapib results in significant increases in HDL-C levels. In hamsters injected with [3H]cholesterol-labeled autologous macrophages Dalcetrapib significantly increases fecal elimination of both [3H]neutral sterols and [3H]bile acids. Dalcetrapib increases plasma HDL-[3H]cholesterol.Dalcetrapib has 95% inhibition of CETP activity in male Japanese white rabbits at an oral dose of 30 mg/kg. Dalcetrapib increases the plasma HDL cholesterol level by 27% and 54%, respectively, when given at oral doses of 30 mg/kg or 100 mg/kg once a day for 3 days to male Japanese white rabbits.Treatment with Dalcetrapib markedly increases serum levels of HDL-C. The ratio of HDL2-C to HDL3-C is significantly higher in Dalcetrapib–treated rabbits than in control rabbits at 5 and 7 months, indicating that the inhibition of CETP activity by Dalcetrapib changes the distribution of HDL subfractions and preferentially increases HDL2-C levels. Dalcetrapib treatment increases serum paraoxonase activity and HDL-associated platelet-activating factor acetylhydrolase activity, but decreases the plasma lysophosphatidylcholine concentration. High-density lipoprotein cholesterol increases associated with co-administration of Dalcetrapib with ezetimibe (+29.8%) are comparable with those with Dalcetrapib alone (+25.6%), while the reduction in low-density lipoprotein cholesterol with co-administration (−35.9%) is greater than with ezetimibe alone (−20.9%). Dalcetrapib is generally well tolerated when administered alone and when co-administered with ezetimibe. The HDL-C increase with Dalcetrapib was not compromised by coadministration with statins, and reduction in low-density lipoprotein cholesterol with Dalcetrapib coadministered with statins was greater than with statins alone. Dalcetrapib alone and coadministered with statins was generally well tolerated. Dalcetrapib has enered in a phase III clinical trial in the treatment of cardiovascular disease, dyslipidemia, peripheral arterial disease (PAD).
| Cell Experiment | |
|---|---|
| Cell lines | HepG2 cells |
| Preparation method | Seeding the HepG2 cells in 6-well plates and cultured to 70–80% confluence. After being washed with PBS, incubating the cells with growth medium and a different concentration (0 μM–30 μM) of chemical inhibitor Dalcetrapib and dissolving in 2% DMSO for 24 hours. Total RNA is used for RT-PCR. |
| Concentrations | 0-30 μM |
| Incubation time | 24 hours |
| Animal Experiment | |
|---|---|
| Animal models | Syrian hamsters |
| Formulation | 0.5% methylcellulose |
| Dosages | 100 mg/kg |
| Administration | Oral gavage |
| Molecular Weight | 389.59 |
| Formula | C23H35NO2S |
| CAS Number | 211513-37-0 |
| Solubility (25°C) | DMSO 70 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
[1] Eric J Niesor, et al. Inhibition of the 3CL Protease and SARS-CoV-2 Replication by Dalcetrapib
[2] Donald M Black, et al. Clinical Pharmacokinetics and Pharmacodynamics of Dalcetrapib
[4] Jennifer G Robinson. Dalcetrapib: a review of Phase II data
| Related CETP Products |
|---|
| Torcetrapib
Torcetrapib (CP-529414) is a prototype cholesteryl ester transfer protein (CETP) inhibitor with potential for decreasing atherosclerotic disease, increased cardiovascular events in clinical trials. |
| Anacetrapib
Anacetrapib is an orally active and potent inhibitor of CETP with IC50 of 7.9 nM and 11.8 nM for rhCETP and mutant CETP(C13S) respectively. |
| Evacetrapib
Evacetrapib (LY2484595) is a potent and selective inhibitor of CETP which inhibited human recombinant CETP protein (5.5 nM IC(50)) and CETP activity in human plasma (36 nM IC(50)) in vitro. |
| Obicetrapib
Obicetrapib (DEZ-001) is a selective, orally active cholesterol transporter protein (CETP) inhibitor that blocks the function of CETP and significantly reduces LDL-C while significantly increasing HDL-C levels, and may be used in studies related to atherosclerosis and Alzheimer's disease (AD). |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.
Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2026 AbMole BioScience. All Rights Reserved.