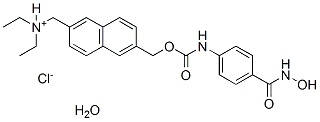
ITF2357 (Givinostat, Gavinostat) is a histone deacetylase inhibitor with potential anti-inflammatory, anti-angiogenic, and antineoplastic activities.The IC50 vale for maize HDAC preparations HD2, HD-1B and HD-1A are 10, 7.5 and 16 nM respectively. ITF2357 (Givinostat, Gavinostat) inhibits class I and class II histone deacetylases (HDACs) and several pro-inflammatory cytokines.This reduces expression of tumour necrosis factor (TNF), interleukin 1α and β, and interleukin 6. ITF2357 (Givinostat,Gavinostat) also has activity against cells expressing JAK (2V617F), a mutated form of the janus kinase 2 (JAK2) enzyme that is implicated in the pathophysiology of many myeloproliferative diseases, including polycythaemia vera.
| Cell Experiment | |
|---|---|
| Cell lines | Purified monocytes |
| Preparation method | Caspase 3/7 Determinations Monocytes were isolated from freshly obtained unfractionated PBMCs by anti-CD14–labeled magnetic beads (CD14 MicroBeads, Miltenyi Biotec, Bergisch Gladbach, Germany) and incubated at 50,000 cells/well in flat-bottom 96-well plates in the presence of increasing concentrations of ITF2357 for 1 h. Thereafter, LPS (10 ng/mL) was added for an additional 24-h incubation. The activity of caspase 3/7 was then determined by Apo-ONE Homogeneous Caspase-3/7 Assay (Promega), and the amount of fluorescence was detected by fluorimetric plate reader (Victor-2; Perkin Elmer). |
| Concentrations | 0~ 1000 nM |
| Incubation time | 24 h |
| Animal Experiment | |
|---|---|
| Animal models | ConA-induced hepatitis in BALB/C mice |
| Formulation | One milligram of ITF2357 was added to 1 mL water and heated to 90° C until dissolved |
| Dosages | 5 mg/kg |
| Administration | gavage |
| Molecular Weight | 475.97 |
| Formula | C24H27N3O4.HCl.H2O |
| CAS Number | 732302-99-7 |
| Solubility (25°C) | DMSO 75 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related HDAC Products |
|---|
| Ac-Arg-Gly-Lys(Ac)-AMC
Ac-Arg-Gly-Lys(Ac)-AMC is a substrate for HDAC. |
| Chlamydocin
Chlamydocin, a fungal metabolite, is a highly potent HDAC inhibitor, with an IC50 of 1.3 nM. |
| HDAC-IN-30
HDAC-IN-30 is a novel multi-target HDAC inhibitor, including HDAC1 (IC50=13.4 nM),HDAC2 (IC50=28.0 nM), HDAC3 (IC50=9.18 nM), HDAC6 (IC50=42.7 nM), HDAC8 (IC50=131 nM). |
| Ac-Arg-Gly-Lys(Ac)-AMC acetate
Ac-Arg-Gly-Lys(Ac)-AMC acetate is a substrate for histone deacetylase (HDAC) and can be used in a novel fluorescent assay for HDAC activity. |
| JPS014 TFA
JPS014 TFA is a benzamide-based Von Hippel-Lindau (VHL) E3-ligase proteolysis targeting chimeras (PROTAC). |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
