All AbMole products are for research use only, cannot be used for human consumption.
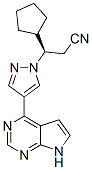
Ruxolitinib (INCB018424) is a first-in-class, potent and selective JAK1/2 inhibitor with an IC50 of 3.3 nM/2.8 nM. It is 130-fold more potent and selective for JAK1 and JAK2 compared to JAK3. In Ba/F3 and HEL cells, INCB018424 potently and selectively inhibited JAK2V617F-mediated signaling and cell proliferation.INCB018424 significantly increased apoptosis in Ba/F3 cells in a dose-dependent manner. In Ba/F3 cells, INCB018424 (64 nM) caused a doubling of mitochondrial depolarized cells.

Cell Death Discov. 2024 Aug 29;10(1):389.
CTDSPL2 promotes the progression of non-small lung cancer through PI3K/AKT signaling via JAK1
INCB18424 purchased from AbMole

Nat Commun. 2023 Nov 27;14(1):7783.
Local H2 release remodels senescence microenvironment for improved repair of injured bone
INCB18424 purchased from AbMole

FASEB J. 2023 Feb;37(2):e22693.
ANGPTL4 inhibits granulosa cell proliferation in polycystic ovary syndrome by EGFR/JAK1/STAT3‐mediated induction of p21
INCB18424 purchased from AbMole

Front Immunol. 2022 May 23;13:866638.
A Novel STAT3 Gain-of-Function Mutation in Fatal Infancy-Onset Interstitial Lung Disease
INCB18424 purchased from AbMole

BMC Biol. 2021 May 20;19(1):108.
Very long intergenic non-coding (vlinc) RNAs directly regulate multiple genes in cis and trans
INCB18424 purchased from AbMole

Blood Adv. 2020 Jul 14;4(13):3000-3010.
Inflammation-driven activation of JAK/STAT signaling reversibly accelerates acute myeloid leukemia in vitro
INCB18424 purchased from AbMole

J Virol. 2020 Feb 14;94(5):e01791-19.
Targeting Kaposi's Sarcoma-Associated Herpesvirus ORF21 Tyrosine Kinase and Viral Lytic Reactivation by Tyrosine Kinase Inhibitors Approved for Clinical Use
INCB18424 purchased from AbMole

Patent. US10335380B2 2019 Jul 02.
Patent. US10335380B2
INCB18424 purchased from AbMole

Patent. JP6479818B2 2019 Mar 06.
Patent. JP6479818B2
INCB18424 purchased from AbMole
| Cell Experiment | |
|---|---|
| Cell lines | Ba/F3-EpoR-JAK2V617F and HEL cell lines |
| Preparation method | Cell proliferation assay. Cells were seeded at 2000/well of white bottom 96-well plates, treated with compounds from DMSO stocks (0.2% final DMSO concentration), and incubated for 48 hours at 37°C with 5% CO2. Viability was measured by cellular ATP determination using the Cell-Titer Glo (Promega) luciferase reagent or viable cell counting. Values were transformed to percent inhibition relative to vehicle control, and IC50 curves were fitted according to nonlinear regression analysis of the data using PRISM GraphPad. |
| Concentrations | 0~10 µ M |
| Incubation time | 48 h |
| Animal Experiment | |
|---|---|
| Animal models | myeloproliferative neoplasm mouse model |
| Formulation | 5% dimethyl acetamide, 0.5% methyl cellulose |
| Dosages | 180mg/kg began within 24 hours of cell inoculation, twice daily |
| Administration | oral gavage |
| Molecular Weight | 306.37 |
| Formula | C17H18N6 |
| CAS Number | 941678-49-5 |
| Solubility (25°C) | DMSO 60 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Related JAK Products |
|---|
| iJak-381
iJak-381 is a JAK1/2 inhibitor with anti-inflammatory activity. |
| Ritlecitinib (malonate)
Ritlecitinib (PF-06651600) malonate is an orally active and selective JAK3 inhibitor with an IC50 of 33.1 nM. |
| Deuruxolitinib
Deuruxolitinib is a deuterated Ruxolitinib (INCB18424), which modulates the activity of JAK1/JAK2. Deuruxolitinib can be used for the research of hair loss disorders. |
| A-005
A-005 is a potentially first-in-class, brain-penetrating TYK2 allosteric inhibitor for studies related to neuroinflammatory and neurodegenerative diseases. |
| Tyrosine Protein Kinase JAK 2 (Phospho-Tyr8, 9)
Tyrosine Protein Kinase JAK 2 (Phospho-Tyr8, 9) is a peptide corresponding to amino acids 475 to 491 of mouse JAK2. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
