All AbMole products are for research use only, cannot be used for human consumption.
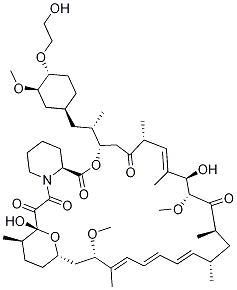
Everolimus (RAD001) also known as SDZ-RAD, Certican, Zortress and Afinitorm is an mTOR inhibitor with IC50 of 0.63 nM. Everolimus is an immunosuppressant analog of rapamycin (sirolimus) and inhibits growth factor-mediated proliferation of haematopoietic and nonhaematopoietic cells. Everolimus shows a potent inhibitory effect on posttransplant lymphoproliferative disorder (PTLD)-derived cells in vitro and in vivo.

Oncogene. 2020 Apr;39(15):3163-3178.
The EGFR-ZNF263 Signaling Axis Silences SIX3 in Glioblastoma Epigenetically
Everolimus purchased from AbMole

Pharmaceutics. 2020 Mar 13;12(3):260.
On-Chip Synthesis of Hyaluronic Acid-Based Nanoparticles for Selective Inhibition of CD44+ Human Mesenchymal Stem Cell Proliferation
Everolimus purchased from AbMole

Gynecol Oncol. 2019 Jun;153(3):661-669.
Chloride channel-3 is required for efficient tumour cell migration and invasion in human cervical squamous cell carcinoma.
Everolimus purchased from AbMole
| Cell Experiment | |
|---|---|
| Cell lines | NCI-N87 and MKN45 cells |
| Preparation method | Proliferation assay. Gastric cancer cells (10,000 cells re-suspended in 1 ml medium) were seeded in 24-well plates. 24 h after seeding,0.5 ml medium containing RAD001 was added to yield desired concentrations of RAD001. At the time points indicated, gastric cancer cells were harvested and counted with a Coulter Z1 electronic cell counter (Beckman Coulter, UK). |
| Concentrations | 2.5–160 n M |
| Incubation time | 72 h |
| Animal Experiment | |
|---|---|
| Animal models | NCI-N87 Tumor xenograft model with Pathogen-free, 4–6 week old, female CB-17 SCID mice |
| Formulation | saline |
| Dosages | 5 mg/kg qd for 121 days |
| Administration | P.O |
| Molecular Weight | 958.22 |
| Formula | C53H83NO14 |
| CAS Number | 159351-69-6 |
| Solubility (25°C) | DMSO 30 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Related mTOR Products |
|---|
| RMC-4627
RMC-4627 is a selective mTORC1 inhibitor that activates 4EBP1 and inhibits tumor growth. |
| RMC-4529
RMC-4529 has an IC50 value of 1.0 nM against p-4E-BP1-(T37/46) in mTOR kinase cellular assay. |
| WYE-687 dihydrochloride
WYE-687 dihydrochloride is an ATP-competitive mTOR inhibitor with an IC50 of 7 nM. |
| Rapamycin-d3
Rapamycin-d3 |
| L-Leucine-d10
L-Leucine-d10 |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
