All AbMole products are for research use only, cannot be used for human consumption.
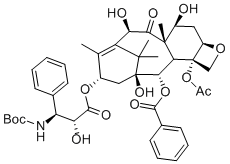
Docetaxel is a clinically well-established anti-mitotic chemotherapy medication. Docetaxel binds to microtubules reversibly with high affinity and has a maximum stoichiometry of 1 mole docetaxel per mole tubulin in microtubules. It has also been found to lead to the phosphorylation of oncoprotein bcl-2, which is apoptosis-blocking in its oncoprotein form. Docetaxel exhibits cytotoxic activity on breast, colorectal, lung, ovarian, gastric, renal and prostate cancer cells. Docetaxel does not block disassembly of interphase microtubules and so does not prevent entry into the mitotic cycle, but does block mitosis by inhibiting mitotic spindle assembly. Docetaxel activity is significantly greater in ovarian and breast tumours than for lung tumours. Docetaxel prevents microtubule depolymerization and disassembly in the absence of GTP.

Mol Med Rep. 2020 Nov;22(5):3935-3943.
Apatinib sensitizes chemoresistant NSCLC cells to doxetaxel via regulating autophagy and enhances the therapeutic efficacy in advanced and refractory/recurrent NSCLC
Docetaxel purchased from AbMole
| Cell Experiment | |
|---|---|
| Cell lines | A549, HCT-116, NCI-H838, KB-3-1, MX-1W, NCI-H1299 and DLD-1 cells |
| Preparation method | Cell Proliferation Assays Cytotoxicity was assessed by growing cells in the presence of agents for 72 h. Cell survival was measured by the sulforhodamine B (SRB) protein stain method or the ATP-binding assay using the Cell-Titer Glo Luminescent Reporter (GLR) System (Promega, Inc., Madison, WI). The SRB assay was done as previously described (23). For the GLR system, cells were plated robotically at approximately 50% confluency in a 384-well plate and allowed to attach for 12 h at 37°C/5% CO2. After diluting test agents using BioMek 2000 robotic system (Beckman Instruments, Fullerton, CA), agents were added to each well and incubated for 72 h. ATP binding and stabilization of the luminescent signal were performed according to manufacturer's protocol (Promega) following tumor cell lysis. Absorbance was read on a Victor V multilabel plate reader (Perkin-Elmer, Gaithersburg, MD) at a wavelength of A595 and data collected using Wallac 1420 Workstation software. |
| Concentrations | 0~100 nM |
| Incubation time | 72 h |
| Animal Experiment | |
|---|---|
| Animal models | Athymic nu/nu female mice bearing Lox melanoma cells, KB-3-1 cells and A375SM melanoma cells tumour xenograft model |
| Formulation | 1.4% ethanol/3.5% polysorbate 80 in saline |
| Dosages | 25 mg/kg |
| Administration | i.v. |
| Molecular Weight | 807.88 |
| Formula | C43H53NO14 |
| CAS Number | 114977-28-5 |
| Solubility (25°C) | DMSO 60 mg/mL Ethanol 50 mg/mL |
| Storage | 2-8°C, protect from light, dry, sealed |
[3] Lyseng-Williamson KA, et al. Drugs. Docetaxel: a review of its use in metastatic breast cancer.
| Related Microtubule Products |
|---|
| Nab-Paclitaxel
Nab-Paclitaxel (Nanoparticle albumin-bound Paclitaxel) is an albumin-bound nanoparticle formulation of Paclitaxel. Nab-Paclitaxel is composed of albumin and the active pharmaceutical ingredient Paclitaxel, in which human albumin is used as an excipient to disperse and stabilize particles and carry the main drug. Nab-Paclitaxel is a solvent-free taxane with higher response rates and improved tolerability. Nab-Paclitaxel displays less toxicity and greater antitumor activity. |
| CCB02
CCB02 is a selective CPAP-tubulin interaction inhibitor, binding to tubulin and competing for the CPAP binding site of β-tubulin, with an IC50 of 689 nM, and shows potent anti-tumor activity. |
| Lisavanbulin
Lisavanbulin (BAL-101553) is the prodrug of the microtubule targeting agent Avanbulin (BAL 27862). |
| 20-O-Demethyl-AP3
20-O-Demethyl-AP3 is a minor metabolite of Ansamitocin P-3. |
| Lisavanbulin dihydrochloride
Lisavanbulin (BAL-101553) dihydrochloride is the prodrug of the microtubule targeting agent Avanbulin (BAL 27862). |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
