10058-F4 inhibits the growth of leukemic cells and dimerization of the c-Myc/Max heterodimerization to control the cell proliferation, apoptosis, and differentiation, and its aberrant expression. 10058-F4 increased FOXO1 mRNA in MedB-1 cells. 10058-F4 is most efficient in inducing neuronal differentiation and lipid accumulation in MYCN-amplified neuroblastoma cells. 10058-F4 could markedly suppresse the wild-type LKB1 loss-induced cell invasiveness. 10058-F4 increased the chemosensitivity of HepG2 cells to low-dose doxorubicin, 5-fluorouracil and cisplatin.

Biochem Pharmacol. 2024 Feb 17;116065.
Panobinostat sensitizes AraC-resistant AML cells to the combination of azacitidine and venetoclax
10058-F4 purchased from AbMole

Cells. 2022 Sep 3;11(17):2752.
Inhibition of Mcl-1 Synergistically Enhances the Antileukemic Activity of Gilteritinib and MRX-2843 in Preclinical Models of FLT3-Mutated Acute Myeloid Leukemia
10058-F4 purchased from AbMole

J Cell Mol Med. 2022 May;26(9):2646-2657.
Venetoclax enhances DNA damage induced by XPO1 inhibitors: A novel mechanism underlying the synergistic antileukaemic effect in acute myeloid leukaemia
10058-F4 purchased from AbMole

Biomed Res Int. 2020 Jun 20;2020:9243681.
Shenmai Injection Supresses Glycolysis and Enhances Cisplatin Cytotoxicity in Cisplatin-Resistant A549/DDP Cells via the AKT-mTOR-c-Myc Signaling Pathway
10058-F4 purchased from AbMole
| Cell Experiment | |
|---|---|
| Cell lines | PC-3 and DU145 cells |
| Preparation method | MTT assay PC-3 cells (2 × 104 cells in logarithmic growth) were plated into 96-well culture plates and allowed to adhere to the plates for 24 h prior to the addition of 10058-F4 in medium containing 1% ethanol such that the final concentrations in the wells were 0.1-100 μM in medium containing 0.3% ethanol. After 72 h, 50 μl of 1 mg/ml MTT was added to each well. The cells were washed with medium and phosphate buffered saline, and 150 μl of DMSO was added to each well, followed by shaking for 5 min. The absorbance at 570 nm was read on DYNEX MRX Revelation microplate reader (Dynex, Vienna, VA, USA). Results were compared to wells containing cells treated with vehicle alone and were expressed as % inhibition. The IC50 was calculated using the Hill equation, the program ADAPT II, and data from three separate experiments. |
| Concentrations | 0.1-100 μM |
| Incubation time | 72 h |
| Animal Experiment | |
|---|---|
| Animal models | SCID mice bearing DU145 or PC-3 xenografts |
| Formulation | cremophor EL:ethanol:saline (1:1:8 v/v/v) at a final concentration of 2 or 3 mg/ml |
| Dosages | 20 or 30 mg/kg daily for 5 days for 2 weeks |
| Administration | i.v. |
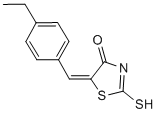
| Molecular Weight | 249.35 |
| Formula | C12H11NOS2 |
| CAS Number | 403811-55-2 |
| Solubility (25°C) | DMSO 30 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
[1] Müller I, et al. PloS one. Targeting of the MYCN Protein with Small Molecule c-MYC Inhibitors.
| Related c-Myc Products |
|---|
| Omomyc
Omomyc is a cell-penetrating peptide derived from the bHLH structural domain of Myc. When expressed in cells, Omomyc inhibits C-Myc-mediated transcription and suppresses cell proliferation.Omomyc competes with C-Myc for binding to Max, resulting in the formation of the Omomyc/Max complex, which competes with the endogenous c-Myc/Max heterodimer and ultimately inhibits transcriptional activation resulting in Myc target genes involved in proliferation and metabolism. |
| MDEG-541
MDEG-541 is a potent MYC-MAX degrader. |
| c-Myc inhibitor 11
c-Myc inhibitor 11 is a c-MYC inhibitor (pEC50: 6.4). |
| c-Myc inhibitor 12
c-Myc inhibitor 12 is an inhibitor of c-Myc with a pEC50 of 6.4. |
| m-Se3
m-Se3 is a potent and selective c-MYC transcription inhibitor that can inhibit tumor growth and has anti-cancer activity. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
