All AbMole products are for research use only, cannot be used for human consumption.
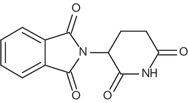
Thalidomide is a sedative and immunomodulator used to study the symptoms of many cancers, it has immunomodulatory, anti-inflammatory and anti-angiogenic cancer properties. Thalidomide inhibits cereblon (CRBN), a part of the cullin-4 E3 ubiquitin ligase complex CUL4-RBX1-DDB1, with a Kd of ∼250 nM.
Thalidomide (50 μg/mL) potentiates the anti-tumor activity of icotinib against the proliferation of both PC9 and A549 cells, and this effect is correlated with apoptosis and cell migration. In addition, Thalidomide and icotinib inhibits the EGFR and VEGF-R2 pathways in PC9 cells.
In vivo, Thalidomide (100 mg/kg, p.o.) inhibits the collagen deposition, down-regulates the mRNA expression level of α-SMA and collagen I, and significantly reduces the pro-inflammatory cytokines in RILF mice. Thalidomide alleviates RILF via suppression of ROS and down-regulation of TGF-β/Smad pathway dependent on Nrf2 status.

Brain Behav Immun. 2022 Feb 7;102:23-39.
Spinal microglia-derived TNF promotes the astrocytic JNK/CXCL1 pathway activation in a mouse model of burn pain
Thalidomide purchased from AbMole

Biomed Pharmacother. 2021 Sep;141:111876.
Cudraxanthone L inhibits gastric cancer by regulating the MAPK signalling and promoting FAS-mediated pathway
Thalidomide purchased from AbMole
| Molecular Weight | 258.23 |
| Formula | C13H10N2O4 |
| CAS Number | 50-35-1 |
| Solubility (25°C) | DMSO 100 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
[1] Takumi Ito, et al. Molecular mechanisms of thalidomide and its derivatives
[2] James E Ridings. The thalidomide disaster, lessons from the past
[3] Michael E Franks, et al. Thalidomide
[4] E Laffitte, et al. Thalidomide
[5] R J D'Amato, et al. Thalidomide is an inhibitor of angiogenesis
| Related Ligand for E3 Ligase Products |
|---|
| OICR-8268
OICR-8268 is a reversible DCAF1 ligand that binds to the DCAF1 WDR domain with a Kd of 38 nM. OICR-8268 can be used in the development of DCAF1-based PROTACs. |
| BC-1215
BC-1215 is a novel inhibitor of F-box protein 3 (Fbxo3) activity. BC-1215 works by antagonizing of Fbxo3 on TRAF cytokine signaling and exhibits a low IC50 in vitro. BC-1215 relieves allodynia by preventing ubiquitination-dependent Fbxl2/TRAF2/TNIK/pGluR1 signaling. BC-1215 exerted robust therapeutic effects in cytokine-driven inflammation, including pseudomona-induced lung injury and cecum ligation/punctured-induced sepsis, by targeting Fbxo3/Fbxl2/TRAF-mediated cytokine release. BC-1215 can be used for the research of inflammation. |
| VH 101, acid
VH 101, acid is a functionalized von-Hippel-Lindau (VHL) protein ligand for PROTAC research and development. |
| (S,R,S)-AHPC-Boc
(S,R,S)-AHPC-Boc (VH032-Boc) is a ligand used in the recruitment of the von Hippel-Lindau (VHL) protein. |
| Lenalidomide-Br
Lenalidomide-Br is an analog of cereblon (CRBN) ligand Lenalidomide for E3 ubiquitin ligase, and is used in the recruitment of CRBN protein. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
