All AbMole products are for research use only, cannot be used for human consumption.
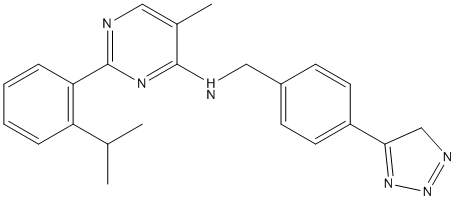
Ml-323 is a potent reversible INHIBITOR of USP1-UAF1, with an IC50 of 76 nM in UB-RHO assay.
| Cell Experiment | |
|---|---|
| Cell lines | H596 cells |
| Preparation method | For the colony-forming assay, seeding cells at a density of 300–500 cells per well in six-well plates and growing overnight.Then treating cells with ML323 alone, cisplatin alone or a combination of cisplatin and ML323 (1:1 or 1:4) at the indicated concentrations. Treating cells with an equal volume of DMSO and usinf saline as control. After 48 h of treatment, adding fresh growth medium, and incubating cells for an additional 5-10 d to allow for colony formation. For UV combination treatment, treating the cells with ML323 at the indicated concentrations or an equal volume of DMSO. After 48 h, removing the medium , and irradiating cells at 254 nm at the indicated dosage. Adding fresh growth medium , and incubating the cells for an additional 5-10 d to allow for colony formation. The cells without UV irradiation but treated with ML323 or an equal volume of DMSO are used as controls and designated as 100%. After the formation of the colonies, fixing cells with methanol and stained with 0.5% crystal violet. Colonies consisting of >50 cells are scored. The number of colonies is determined from triplicate plates. The dose-response curves are generated using GraphPad Prism and analyzed by using CalcuSyn to calculate the combination index, which is determined for the fraction of cells affected after the addition of fixed ratios of cisplatin and the USP1-UAF1 inhibitor. |
| Concentrations | ~30 μM |
| Incubation time | 7-12 days |
| Animal Experiment | |
|---|---|
| Animal models | |
| Formulation | |
| Dosages | |
| Administration | |
| Molecular Weight | 384.48 |
| Formula | C23H24N6 |
| CAS Number | 1572414-83-5 |
| Solubility (25°C) | DMSO |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Related Dehydrogenase Products |
|---|
| MTHFD2-IN-4
MTHFD2-IN-4, tricyclic coumarin derivative, is a potent MTHFD2 inhibitor. |
| MTHFD2-IN-4 sodium
MTHFD2-IN-4 sodium, tricyclic coumarin derivative, is a potent MTHFD2 inhibitor. |
| MTHFD2-IN-3
MTHFD2-IN-3 is a potent methylenetetrahydrofolate dehydrogenase (MTHFD2) inhibitor. |
| MTHFD2-IN-2
MTHFD2-IN-2 is a potent methylenetetrahydrofolate dehydrogenase (MTHFD2) inhibitor. |
| MTHFD2-IN-1
MTHFD2-IN-1 is a potent methylenetetrahydrofolate dehydrogenase (MTHFD2) inhibitor. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
