All AbMole products are for research use only, cannot be used for human consumption.
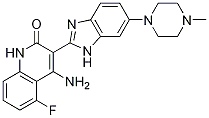
Dovitinib (TKI258) potently inhibited FLT3, c-KIT, FGFR, VEGFR1/2/3, PDGFRß and CSF-1R with IC50 values of 1, 2, 5, 10, 8, 27, 36 nM respectively. Dovitinib selectively blocked the growth of wild-type (WT) or activated mutant FGFR3-transformed B9 cells and human myeloma cell lines. Dovitinib was an effective treatment in a xenograft mouse model of FGFR3 multiple myeloma.
| Cell Experiment | |
|---|---|
| Cell lines | J807C, Y373C, K650E, G384D, wild type (WT), F384L and B9-MINV cells |
| Preparation method | Viability assay. Cell viability was assessed by 3-(4,5-dimethylthiazol)-2,5-diphenyl tetrazolium (MTT) dye absorbance according to the manufacturer's instructions (Boehringer Mannheim, Mannheim, Germany). Cells were seeded in 96-well plates at a density of 5000 (B9 cells) or 20 000 (MM cell lines) cells per well. Cells were incubated with 30 ng/mL aFGF and 100 μg/mL heparin or 1% IL-6 where indicated and increasing concentrations of CHIR-258. For each concentration of CHIR-258, 10-μL aliquots of drug or DMSO diluted in culture medium was added. For drug combination studies, cells were incubated with 0.5 μM dexamethasone, 100 nM CHIR-258, or both simultaneously where indicated. To evaluate the effect of CHIR-258 on growth of MM cells adherent to BMSCs, 10 000 KMS11 cells were cultured on BMSC-coated 96-well plates in the presence or absence of CHIR-258. Plates were incubated for 48 to 96 hours. |
| Concentrations | 0~400 n M |
| Incubation time | 48 h |
| Animal Experiment | |
|---|---|
| Animal models | KMS11 cells xenograft mouse model |
| Formulation | 5 mM citrate buffer |
| Dosages | 10, 30, or 60 mg/kg daily for 21 days |
| Administration | oral gavage |
| Molecular Weight | 392.43 |
| Formula | C21H21FN6O |
| CAS Number | 405169-16-6 |
| Solubility (25°C) | DMSO 30 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Related VEGFR/PDGFR Products |
|---|
| SU5208
SU5208 inhibits vascular endothelial growth factor receptor-2 (VEGFR2). |
| VEGFR-IN-1
VEGFR-IN-1 is a potent angiogenesis inhibitor with IC50s of 0.02, 0.18, 0.24 7.3, and 7 µM for KDR, Flt-1, c-Kit, EGF-R, and c-Src, respectively. |
| (Z)-Orantinib
(Z)-Orantinib ((Z)-SU6668) is a potent, selective, orally active and ATP competitive inhibitor of Flk‐1/KDR, PDGFRβ, and FGFR1, with IC50s of 2.1, 0.008, and 1.2 µM, respectively. |
| (Rac)-SAR131675
(Rac)-SAR131675 is the racemate of SAR131675. |
| AGL 2043
AGL 2043 is an effective inhibitor of PDGFR (IC50=0.8 μM) and TEL-PDGFR kinases, as well as FLT3 and KIT kinases. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
