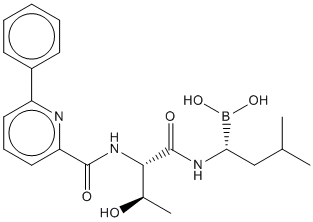
Delanzomib (CEP-18770) is a potent chymotrypsin-like proteasome inhibitor with an IC50 of 3.8 nM. Delanzomib (CEP-18770) prevents the proliferation of several tumor cell lines, such as A2780 ovarian cancer cells, PC3 prostate cancer, H460, LoVo colon cancer, RPMI8226 multiple myeloma cancer and HS-Sultan anaplastic non-Hodgkin lymphoma with IC50 of 13.7, 22.2, 34.2 11.3, 5.6 and 8.2 nM, respectively. The IC50 values of Delanzomib (CEP-18770) are similar to those of bortezomib, with the chymotryptic and caspase-like activities being inhibited at low-nanomolar concentrations. Delanzomib (CEP-18770) blocks the ubiquitin-proteasome pathway in several MM and in the chronic myelogenous leukemia cell line, K562. Delanzomib (CEP-18770) gives rise to an accumulation of ubiquitinated proteins over 4 to 8 hours with a profile similar to that observed after bortezomib treatment.Delanzomib (CEP-18770) completely blocks the degradation of IκBα. Delanzomib (CEP-18770) markedly decreases high levels of NF-κB activity in both RPMI-8226 and U266 cells. The time- and concentration-dependent suppression of NF-kB DNA-binding activity in MM cell lines by Delanzomib (CEP-18770) leads to a decrease of several NF-κB-modulated genes mediating the growth and survival of tumor cells including IkBα itself, the X-chromosome-linked inhibitor-of-apoptosis protein (XIAP), the pro-inflammatory cytokines TNF-α and interleukin-1β (IL-1β), the intracellular adhesion molecule (ICAM1), and the pro-angiogeneic factor vascular endothelial growth factor. The proapoptotic activity of Delanzomib (CEP-18770) against MM is not limited solely to tumor-derived MM cell lines, but extends to primary MM explants from relapsed or refractory patients including those previously treated with bortezomib. In addition, the combination of Delanzomib (CEP-18770) with melphalan or bortezomib generates synergistic prevention of MM cell viability in vitro. Delanzomib (CEP-18770) reveals sustained and dose-related relative tumor weight inhibition. Delanzomib (CEP-18770) leads to dose-related induction of complete tumor regressions, as compared with bortezomib treatment, which results in a 50% incidence of CR at its maximally tolerated dose (MTD) of 1.2 mg/kg intravenously.In contrast to bortezomib, Delanzomib (CEP-18770) reveals dose-related increases in the incidence of tumor-free mice by the completion of these studies (120 days after tumor transplantation). Oral administration of Delanzomib (CEP-18770) yields a marked decrease of tumor weight and notable dose-related incidence of complete tumor regression with minimal changes in animal body weight over the course of 120 day studies. Relative to bortezomib, equiactive doses of Delanzomib (CEP-18770) reveals a greater and more sustained dose-related inhibition of tumor proteasome activity, corresponding temporally with maximum induction of caspase-3 and 7 activity.The maximum apoptotic signal is 2.5 fold greater for Delanzomib (CEP-18770) versus bortezomib.
| Cell Experiment | |
|---|---|
| Cell lines | HMEC and TEC cells |
| Preparation method | Seeding HMEC and TEC cells into 24-well plates at a density of 104 cells/well in DMEM supplemented with 5% FCS. After incubation with proteasome inhibitors (48 hours), cells are washed, air dried, and stained with crystal violet as described. In duplicate samples,cell number is determined on the basis of a standard curve obtained with known cell numbers. All experiments are performed in triplicate. In vitro formation of capillary-like structures is studied on cells (4 × 104 cells/well in DMEM supplemented with 5% FCS. After incubation with proteasome inhibitors (48 hours), washing cells (cells/well in 24-well plates) and seeding onto Matrigel-coated wells in DMEM containing 0.25% BSA. HMEC and TEC cells (5 × 103 per well), suspended in 200 μL DMEM with 5% FCS (positive control), serum-free medium (negative control), are layered onto the Matrigel surface in the presence or absence of proteasome inhibitor CEP-18770. Cells are observed with a inverted microscope and experimental results are then recorded after a 6-hour incubation at 37 °C. Data is analyzed, as the mean (× 1 SD) of total length of capillary-like structures, by the Micro-Image system and is expressed as mm/field by the computer analysis system in 5 different fields at 100 × magnification in duplicated wells for 4 different experiments. |
| Concentrations | 0-100 nM |
| Incubation time | 6 hours |
| Animal Experiment | |
|---|---|
| Animal models | Human MM RPMI 8226 subcutaneous xenograft model in SCID mice |
| Formulation | In a solution of 3% DMSO |
| Dosages | From 1.5 to 4 mg/kg, twice for 7 days to 4 weeks. |
| Administration | Intravenously |
| Molecular Weight | 413.28 |
| Formula | C21H28BN3O5 |
| CAS Number | 847499-27-8 |
| Solubility (25°C) | DMSO 73 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related Proteasome Products |
|---|
| NIC-0102
NIC-0102 is an orally potent proteasome inhibitor (pIC50=7.55) that specifically inhibits NLRP3 inflammasome activation.NIC-0102 showed potent anti-inflammatory effects in vivo in a DSS-induced ulcerative colitis model. In addition, NIC-0102 inhibited the production of pro-IL-1β. |
| Ac-WLA-AMC
Ac-WLA-AMC is a specific 20S constitutive proteasome β5 fluorogenic substrate. |
| Ac-Nle-Pro-Nle-Asp-AMC
Ac-Nle-Pro-Nle-Asp-AMC is a specific substrate for 26S proteasome. |
| PR-39
PR-39, a natural proline- and arginine-rich antibacterial peptide, is a noncompetitive, reversible and allosteric proteasome inhibitor. |
| PSI
PSI (Proteasome Inhibitor 1) is a potent proteasome inhibitor. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
