All AbMole products are for research use only, cannot be used for human consumption.
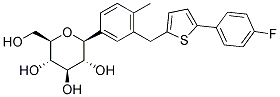
Canagliflozin is an inhibitor of subtype 2 sodium-glucose transport protein (SGLT2) for CHO-hSGLT2, CHO-rSGLT2 and CHO-mSGLT2 with IC50 of 4.4 nM, 3.7 nM and 2 nM, which is responsible for at least 90% of the glucose reabsorption in the kidney. Canagliflozin increased LDL cholesterol, urinary tract infections, genital mycotic infections, and was associated increased urination and episodes of hypotension and hypoglycemia. Canagliflozin shows pronounced anti-hyperglycemic effects in high-fat diet fed KK (HF-KK) mice. Oral administration at 30 mg/kg of Canagliflozin to male SD rats induces glucose excretion over 24 hours by 3,696 mg per 200 g body weight. Dose dependently, canagliflozin decreased calculated 24-h mean RT(G) with maximal reduction to approximately 60 mg/dl, and increased mean 24-h UGE. At doses >200 mg administered before breakfast, canagliflozin reduced postprandial PG and serum insulin excursions at that meal. Canagliflozin was generally well tolerated. Canagliflozin has entered in a Phase III clinical trial for the treatment of diabetes mellitus and type 2 renal insufficiency.
| Molecular Weight | 444.52 |
| Formula | C24H25FO5S |
| CAS Number | 842133-18-0 |
| Solubility (25°C) | DMSO ≥ 50 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Related SGLT Products |
|---|
| Velagliflozin
Velagliflozin is an orally available sodium-glucose cotransporter 2 (SGLT2) inhibitor, with anti-diabetic activity. |
| Tianagliflozin
Tianagliflozin is an SGLT2 inhibitor that may be used in studies related to type 2 diabetes. |
| JTT-662
JTT-662 is an SGLT1 inhibitor that can be used in studies related to type 2 diabetes. |
| JP-2266
JP-2266 is a dual SGLT1/2 inhibitor that can be used in studies related to diabetes. |
| Tofogliflozin
Tofogliflozin(CSG-452) is a potent and highly specific sodium/glucose cotransporter 2(SGLT2) inhibitor with Ki values of 2.9, 14.9, and 6.4 nM for human, rat, and mouse SGLT2. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
