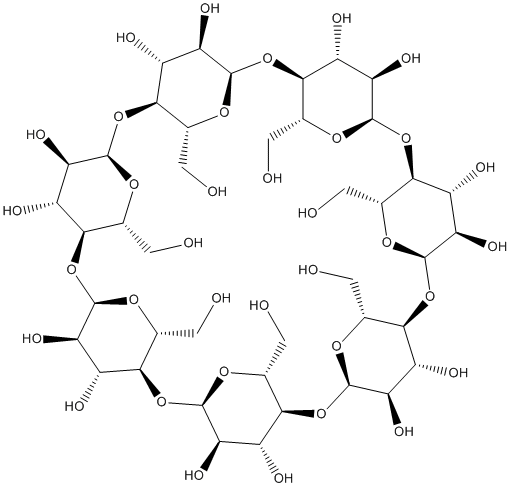
β-Cyclodextrin affects the formation of isomerization products during peptide deamidation.
J Exp Med. 2017 May 1;214(5):1387-1409.
Liver carcinogenesis by FOS-dependent inflammation and cholesterol dysregulation
β-Cyclodextrin purchased from AbMole
| Molecular Weight | 1134.98 |
| Formula | C42H70O35 |
| CAS Number | 7585-39-9 |
| Form | Solid |
| Solubility (25°C) | DMSO |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related Anti-infection Products |
|---|
| Sulfogaiacol
Sulfogaiacol is a antitussive agent. Sulfogaiacol is used for acute respiratory tract infections, cough and other conditions. |
| 2,4-Dichlorobenzyl alcohol
2,4-Dichlorobenzyl alcohol is a mild antiseptic, with a broad spectrum for bacterial and virus associated with mouth and throat infections. |
| 1,2,3,4-Tetrahydronorharman-1-one
1,2,3,4-Tetrahydronorharman-1-one is a manzamine alkaloid with activity against infectious and tropical parasitic diseases from an Indonesian sponge. |
| Acetylalkannin
Acetylalkannin (Alkannin acetate) is an isohexenylnaphthazarin pigment isolated from Arnebia euchroma with antimicrobial and cytotoxic activities. |
| Chimeric Rabies Virus Glycoprotein Fragment (RVG-9R)
Chimeric Rabies Virus Glycoprotein Fragment (RVG-9R), a chimeric peptide consisting of 29 amino acids, is synthesized by adding nona-arginine motif to the carboxy terminus of RVG (rabies virus glycoprotein). |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
