All AbMole products are for research use only, cannot be used for human consumption.
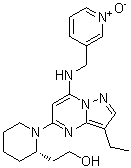
SCH727965 (Dinaciclib) is a novel small molecule multi-CDK inhibitor with low nanomolar potency against CDK1, CDK2, CDK5 and CDK9 that has shown favorable toxicity and efficacy in preliminary mouse experiments, and has been well tolerated in Phase I clinical trials. Cyclin-dependent kinases (CDK) are key positive regulators of cell cycle progression and attractive targets in oncology. SCH727965 was selected as a clinical candidate using a functional screen in vivo that integrated both efficacy and safety parameters. Compared with flavopiridol, SCH727965 exhibits superior activity with an improved therapeutic index. In cell-based assays, SCH727965 completely suppressed retinoblastoma phosphorylation, which correlated with apoptosis onset and total inhibition of bromodeoxyuridine incorporation in >100 tumor cell lines of diverse origin and background. Moreover, short exposures to SCH727965 were sufficient for long-lasting cellular effects. SCH727965 induced regression of established solid tumors in a range of mouse models following intermittent scheduling of doses below the maximally tolerated level. These results suggest that SCH727965 is a potent and selective CDK inhibitor and a novel cytotoxic agent.

Clin Cancer Res. 2018 Nov 15;24(22):5658-5672.
Increased Synthesis of MCL-1 Protein Underlies Initial Survival of EGFR-Mutant Lung Cancer to EGFR Inhibitors and Provides a Novel Drug Target.
SCH727965 (dinaciclib) purchased from AbMole
| Cell Experiment | |
|---|---|
| Cell lines | A2780 cells |
| Preparation method | dThd uptake growth inhibition assay. A2780 cells were maintained in DMEM (Cellgro) plus 10% fetal bovine serum (HyClone) and passaged twice weekly by detaching the monolayer with trypsin-EDTA (Life Technologies). One hundred microliters of A2780 cells (5 × 103 cells) were added per well to a 96-well Cytostar-T plate (Amersham) and incubated for 16 to 24 hours at 37°C. Compounds were serially diluted in complete media plus 2% 14C-labeled dThd (Amersham). Media were removed from the Cytostar T plate; 200 μL of various compound dilutions were added in quadruplicate; and the cells were incubated for 24 hours at 37°C. Accumulated incorporation of radiolabel was assayed using scintillation proximity and measured on a TopCount (Packard/Perkin-Elmer Life Sciences). The percentage of dThd uptake inhibition, relative to a vehicle control, was calculated and plotted on log-linear plots to allow derivation of IC50 values. |
| Concentrations | 0~500nM |
| Incubation time | 16 to 24 h |
| Animal Experiment | |
|---|---|
| Animal models | A2780 cells xenograft model in mice |
| Formulation | 20% hydroxypropyl-β-cyclodextran |
| Dosages | 8, 16, 32, and 48 mg/kg daily for 10 d |
| Administration | i.p. |
| Molecular Weight | 396.48 |
| Formula | C21H28N6O2 |
| CAS Number | 779353-01-4 |
| Solubility (25°C) | DMSO 55 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Related CDK Products |
|---|
| (E/Z)-Zotiraciclib citrate
(E/Z)-Zotiraciclib citrate is a potent CDK2, JAK2, and FLT3 inhibitor. |
| Abemaciclib metabolites M2
Abemaciclib metabolite M2 (LSN2839567) is a metabolite of Abemaciclib, acts as a potent CDK4 and CDK6 inhibitor, with IC50 values of 1.2 and 1.3 nM, respectively. |
| CDK9-IN-1
CDK9-IN-1 is a novel, selective CDK9 inhibitor, with an IC50 of 39 nM for CDK9/CycT1, it can be used for the research of HIV infection. |
| XO44
XO44 (PF-6808472) is a broad-spectrum covalent kinase probe. |
| ML 315
ML 315 is a selective dual inhibitor of CDK and DYRK with IC50s of 68 nM and 282 nM, respectively. |
All AbMole products are for research use only, cannot be used for human consumption or veterinary use. We do not provide products or services to individuals. Please comply with the intended use and do not use AbMole products for any other purpose.


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2024 AbMole BioScience. All Rights Reserved.
