In vitro: YM-58483 can decrease the levels of P-ERK and P-CREB, without affecting the expression of CD11b and GFAP. YM-58483 also inhibits the release of spinal cord IL-1β, TNF-α, and PGE2. YM-58483 and cyclosporine A inhibits T cell proliferation in a one-way mixed lymphocyte reaction (mLR) with IC50 values of 330 and 12.7 nM, respectively. YM-58483 inhibits DNP antigen-induced histamine release from and leukotrienes (LTs) production in IgE-primed RBL-2H3 cells, a rat basophilic leukemia cell line, with IC50 values of 460 and 310 nM, respectively. YM-58483 also inhibits phytohemagglutinin-P (PHA)-stimulated IL-5 and IL-13 production in human peripheral blood cells with IC50 values of 125 and 148 nM, respectively, which is approximately 5 times less potent than prednisolone. YM-58483 inhibits IL-4 and IL-5 production in a conalbumine-stimulated murine Th2 T cell clone (D10.G4.1), and IL-5 production in phytohemagglutinin-stimulated human whole blood cells with IC50 values comparable to those reported for its CRAC channel inhibition (around 100 nM).
In vivo: Intrathecal YM-58483 at the concentration of 300 μM (1.5 nmol) and 1000 μM (10 nmol) produces a significant central analgesic effect on the SNL rats. In the mouse graft-versus-host disease (GVHD) model, YM-58483 (1-30 mg/kg, p.o.) and cyclosporine A (1-30 mg/kg, p.o.) inhibit donor anti-host cytotoxic T lymphocyte (CTL) activity and IFN-γ production, and also reduce the number of donor T cells, especially donor CD8+ T cells, in the spleen. YM-58483 (1-10 mg/kg, p.o.) and cyclosporine A (2, 10 mg/kg, p.o.) inhibit the sheep red blood cell (SRBC)-induced delayed type hypersensitivity (DTH) response. M-58483 (30 mg/kg, p.o.) significantly suppresses ovalbumin (OVA)-induced bronchoconstriction in OVA-sensitized guinea pigs, whereas prednisolone does not. YM-58483 (3-30 mg/kg, p.o.) and prednisolone (100 mg/kg, p.o.) both significantly and completely suppress airway hyperresponsiveness (AHR) caused by OVA exposure. YM-58483 inhibits antigen-induced eosinophil infiltration into airways, and decreases IL-4 and cysteinyl-leukotrienes content in inflammatory airways induced in actively sensitized Brown Norway rats. Orally administered YM-58483 prevents antigen-induced late phase asthmatic broncoconstriction and eosinophil infiltration in actively sensitized guinea pigs.
| Cell Experiment | |
|---|---|
| Cell lines | RPE cell line ARPE19 |
| Preparation method | Cells were pre-treated with MRS1845 (10 μM), YM-58483 (10 μM), xestospongin C (1 μM) or caffeine (10 mM) for 30 min, and then incubated with 300 lg/ml MGO for 3 h. |
| Concentrations | 10 μM |
| Incubation time | 30 min |
| Animal Experiment | |
|---|---|
| Animal models | Male Balb/c mice |
| Formulation | 0.5% methylcellulose solution |
| Dosages | 10 mL/k |
| Administration | oral |
| Molecular Weight | 421.32 |
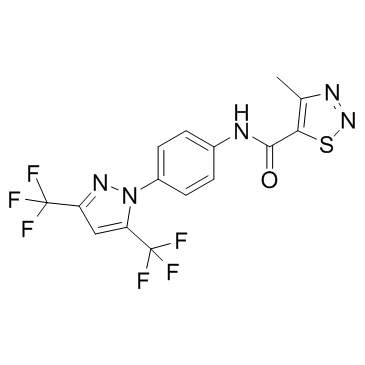
| Formula | C15H9F6N5OS |
| CAS Number | 223499-30-7 |
| Solubility (25°C) | 32 mg/mL in DMSO |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related Calcium Channel Products |
|---|
| ω-Agatoxin TK
ω-Agatoxin TK, a peptidyl toxin of the venom of Agelenopsis aperta, is a potent and selective P/Q type Ca2+ channel blocker. |
| SNX-482
SNX-482, a peptidyl toxin of the spider Hysterocrates gigas, is a potent, high affinity, selective and voltage-dependent R-type CaV2.3 channel blocker with an IC50 of 30 nM. |
| ω-Conotoxin MVIIC
ω-Conotoxin MVIIC is a N- and P/Q-type Ca2+ channel blocker, significantly suppresses the 11-keto-βboswellic acid-mediated inhibition of glutamate release. |
| ω-Conotoxin GVIA
ω-Conotoxin GVIA is an inhibitor of the N-type Ca2+ channel. |
| ω-Conotoxin CnVIIA
ω-Conotoxin CnVIIA, a 27 amino acid neuropeptide toxin, is a N-type calcium current blocker. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
