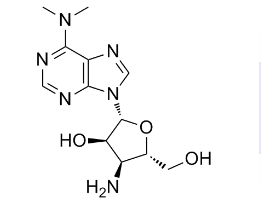
Puromycin aminonucleoside (PAN), which is an analogue of puromycin antibiotic, has been largely adopted to experimentally induce MCD in rodents (with a single high dose) or FSGS (by repeated doses for over a longer period).
In vitro: Puromycin aminonucleoside (30 μg/mL) markedly increases p53 protein levels in podocytes. Puromycin aminonucleoside-induced podocyte apoptosis is p53 dependent and supports the notion that dexamethasone exerts an antiapoptotic effect on cells that are exposed to Puromycin aminonucleoside through the downregulation of p53. Puromycin aminonucleoside induces podocyte apoptosis in a time-dependent manner. The IC50 values for PMAT-expressing and vector-transfected cells are 48.9 and 122.1 μM, respectively, suggesting expression of PMAT-enhanced cell sensitivity to Puromycin aminonucleoside. Puromycin aminonucleoside (250 μM) is toxic to both PMAT-expressing and vector-transfected cells. Puromycin aminonucleoside uptake in PMAT-expressing cells is fourfold higher at pH 6.6 than that at pH 7.4.
In vivo: The number of podocytes per glomerulus is 95.5±17.6 in the control rats, 90.7 on Day 4 in Puromycin aminonucleoside (8 mg/100 g, i.v.)-treated nephrosis rats. The amount of nephrin per glomerulus in control rats is 1.02±0.11 fmol and those in Puromycin aminonucleoside nephrosis rats are reduced to 0.46±0.06 fmol and 0.35±0.04 fmol on Day 4 and Day 7. The nephrin amount per podocyte is significantly decreased association with the development of proteinuria in Puromycin aminonucleoside nephrosis rats. Rats given Puromycin aminonucleoside (100 mg/kg, s.c.) gain less weight and their serum creatinine levels are higher than the control rats, indicating imPuromycin aminonucleosideired renal function.
| Cell Experiment | |
|---|---|
| Cell lines | The human podocyte cell line |
| Preparation method | Cells are seeded in MEM with 10% FBS on 96-well plates at a density of 5,000 cells/well. After appr 48-h incubation (appr 40-50% confluence), cells are changed to fresh growth medium containing Puromycin aminonucleoside at various concentrations. For the protection experiment, cells are incubated in medium containing 250 μM Puromycin aminonucleoside with or without the PMAT inhibitor decynium-22 (2 μM). After a total of 72-h incubation in a 95% O2 incubator at 37°C, cells are washed and the plates. |
| Concentrations | 250 μM |
| Incubation time | 72 h |
| Animal Experiment | |
|---|---|
| Animal models | Male F344 rats |
| Formulation | saline |
| Dosages | 8 mg/100 g body weight |
| Administration | i.v. |
| Molecular Weight | 294.31 |
| Formula | C12H18N6O3 |
| CAS Number | 58-60-6 |
| Solubility (25°C) | Water 30 mg/mL DMSO 15 mg/mL |
| Storage | -20°C, protect from light |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related Apoptosis Products |
|---|
| Cytochrome C (from Pig Heart)
Cytochrome C (from Pig Heart) is a multifunctional enzyme involved in cellular life and death decisions.Cytochrome C is critical in mitochondrial electron transport and intrinsic type II apoptosis. |
| Celosin K
Celosin K, a triterpenoid saponin isolated from the seeds of Semen Celosiae, is also a potent inhibitor of t-BHP-induced neuronal damage. In addition, Celosin K inhibits oxidative stress, apoptosis, and autophagy. |
| GLP-2(rat)
GLP-2(rat) is an intestinal growth factor. |
| Edratide
Edratide (TV 4710) is a synthetic peptide of 19 amino acid based on the complementarity-determining region 1 (CDR1) of a human anti-DNA antibody that expresses a major idiotype denoted 16/6 Id. |
| Thrombospondin-1 (1016-1023) (human, bovine, mouse)
Thrombospondin-1 (1016-1023) (human, bovine, mouse), is the C-terminal end of the native sequence of Thrombospondin-1 (TSP-1), is a CD47 agonist peptide. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
