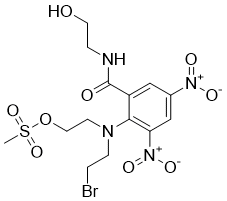
PR-104A is a corresponding alcohol that is systemically converted from PR-104 and a hypoxia-targeted anticancer agent with IC50 of 153 and 60 μM for wild-type CHO AA8 under aerobic and anoxic conditions. PR-104A itself showed modest but significant selectivity for anoxic cells. In a study, the estimated unbound human clearance of PR-104A was 211 L/h/70 kg, with a steady state unbound volume of 105 L/70 kg. The size equivalent unbound PR-104A clearance was 2.5 times faster in dogs, 0.78 times slower in rats and 0.63 times slower in mice, which may reflect reported species differences in PR-104A O-glucuronidation. PR-104A is glucuronidated by UGT2B7 with high specificity and appears to make a major contribution to clearance of PR-104A in humans.
| Molecular Weight | 514.33 |
| Formula | C15H22BrN4O9S |
| CAS Number | 680199-06-8 |
| Solubility (25°C) | DMSO |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related DNA/RNA Synthesis Products |
|---|
| Alternariol monomethyl ether
Alternariol monomethyl ether is an alternaria mycotoxin and genotoxin, found in common edible crops. It inhibits the activity of various DNA-topoisomerases, increasing the rate of DNA strand breaks. |
| APE1-IN-1
APE1-IN-1 is a potent and blood-brain barrier (BBB) penetrant apurinic/apyrimidinic (AP) endonuclease 1 (APE1) inhibitor with an IC50 value of 2 μM. APE1-IN-1 can potentiate the cytotoxicity of the alkylating agents Methylmethane sulfonate to cancer cells. |
| DTP3 TFA
DTP3 TFA is a potent and selective GADD45β/MKK7 (growth arrest and DNA-damage-inducible β/mitogen-activated protein kinase kinase 7) inhibitor. |
| Mating Factor α (1-6)
Mating Factor α (1-6) is a mating factor produced by α-mating type cells of Saccharomyces cerevisiae and an acts as an inhibitor of the initiation of DNA synthesis in the cells. |
| Polynucleotide kinase
Polynucleotide Kinase is a DNA repair enzyme. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
