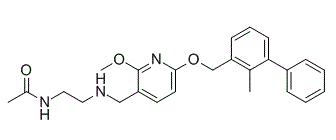
Bms-202 is an effective non-peptide PD-1/PD-L complex inhibitor with IC50 of 18 nM and KD of 8 μM. Bms-202 directly binds to PD-L1 and blocks the human PD-1/PD-L interaction. Bms-202 has antitumor activity.
| Molecular Weight | 419.52 |
| Formula | C25H29N3O3 |
| CAS Number | 1675203-84-5 |
| Solubility (25°C) | DMSO: ≥ 40 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related PD-1/PD-L1 Products |
|---|
| Recombinant Anti-Mouse PD1 mAb
Recombinant Anti-Mouse PD1 mAb is a recombinant monoclonal antibody against the mouse PD1 protein, with the antibody isoform Mouse lgG1/lambda. the antibody efficiently binds to the mPD1 protein on the cell surface and significantly inhibits tumor growth in a mouse tumor model transplanted with MC38 cells. the antibody is used to detect the presence of the mouse PD1 protein on the cell surface of the cell. the antibody is used to detect the presence of the mouse PD1 protein on the cell surface of the cell. |
| Erfonrilimab
Erfonrilimab is a humanized single-domain bispecific antibody-Fc fusion protein (VH-VH-h-CH2-CH3 dimer) targeting the immune checkpoints PD-L1 and CTLA-4. |
| Benmelstobart
Benmelstobart is an anti-PD-L1 humanized IgG1ҡ monoclonal antibody that can be used for tumor-related studies. |
| Human PD-L1 inhibitor II
Human PD-L1 inhibitor II is a potent PD-L1 inhibitor with anti-cancer activity. |
| Human PD-L1 inhibitor I
Human PD-L1 inhibitor I is a hPD-1 peptide ligand, with a KD of 3.39 μM. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
