INO-1001 is a novel poly(ADP-ribose) polymerase (PARP) inhibitor improves cardiac and pulmonary function after crystalloid cardioplegia and extracorporal circulation. The present study tested INO-1001 for its in vivo effect on the chemoresponse of two p53 deficient tumors, human breast cancer MDA-MB-231 and murine mammary carcinoma MCa-K. Doxorubicin was used as the DNA damaging agent and tumor growth delay assay was used as the endpoint. Results showed that INO-1001 was highly effective in enhancing the anti-tumor effects of Doxorubicin for both MDA-MB-231 (EF=1.88) and MCa-K (EF=1.64).

Bioact Mater. 2022 Feb 9.
pH and lipase-responsive nanocarrier-mediated dual drug delivery system to treat periodontitis in diabetic rats
INO-1001 purchased from AbMole

Cell Commun Signal. 2020 Feb 17;18(1):27.
mTOR May Interact With PARP-1 to Regulate Visible Light-Induced Parthanatos in Photoreceptors
INO-1001 purchased from AbMole
| Cell Experiment | |
|---|---|
| Cell lines | cultured podocytes |
| Preparation method | Apoptosis detection. Apoptotic nuclei of cultured podocytes were detected on paraformaldehyde-fixed cells using DAPI (4′,6-diamidino-2-phenylindole) staining (1 μg/ml) for 10 min. Cells were analyzed under a fluorescence microscope and assessed for chromatin condensation and segregation. Caspase-3 activity was measured in podocyte extracts using EnzCheck Caspase-3 Assay kit (Molecular Probes) following the manufacturer’s protocol. Nuclear extracts were prepared using Transfactor DB Nuclear extraction kit (Clontech BD Bioscience) following the manufacturer’s protocol. Western blotting was performed using NFκB p50 (Santa Cruz Biotechnology) antibody. NFκB p50 nuclear binding assay was performed using TransFactor Colorimetric kit (Clontech BD) according to the manufacturer’s protocol. |
| Concentrations | 200 nmol/l |
| Incubation time | 45 min |
| Animal Experiment | |
|---|---|
| Animal models | diabetic nephropathy of Leprdb/db mice model |
| Formulation | sterile water |
| Dosages | 60 mg/kg initiated at 5 weeks of age to around 8 weeks of age |
| Administration | oral in drinking water |
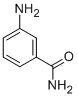
| Molecular Weight | 136.15 |
| Formula | C7H8N2O |
| CAS Number | 3544-24-9 |
| Solubility (25°C) | DMSO 22 mg/mL Water ≥ 4 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related PARP Products |
|---|
| DSB-1522
DSB-1522 is a PARP1 inhibitor that can be used in tumor-related studies. |
| DM-5167
DM-5167 is a PARP1 inhibitor that can be used in studies related to triple negative breast cancer. |
| NMS-03305293
NMS-03305293 is a PARP inhibitor with high selectivity for PARP1 isoforms and low DNA capture effect, which specifically kills BRCA mutated tumor cells. In addition, NMS-03305293 can penetrate the blood-brain barrier and can be used in studies related to CNS tumors and brain metastatic tumors. |
| SNV1521
SNV1521 is a potential best-in-class (BIC), highly selective and CNS-permeable PARP1 inhibitor. |
| Basroparib
Basroparib is an orally active selective inhibitor of end-anchor polymerase (TNKS) and an inhibitor of ribose polymerase (PARP) with antitumor activity. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
