Dacomitinib (PF-00299804) is an oral, irreversible, small molecule inhibitor of human epidermal growth factor receptor-1, -2, and -4 tyrosine kinases. PF00299804 is a potent inhibitor of EGFR-activating mutations as well as the EGFR T790M resistance mutation both in vitro and in vivo. The human EGF (HER) family of receptors has been pursued as therapeutic targets in breast cancer and other malignancies. Dacomitinib inhibited growth in several HER2-amplified lines with de novo and acquired resistance to trastuzumab. Dacomitinib was administered orally at three dose levels (15, 30, or 45 mg once daily [QD]). Single-dose administration of dacomitinib plus DM was safe and well tolerated in HVs and resulted in a significant increase in systemic exposures of DM in extensive metabolizers. Additionally, PF00299804 is a highly effective inhibitor of both the wild-type ERBB2 and the gefitinib-resistant oncogenic ERBB2 mutation identified in lung cancers.

Clin Cancer Res. 2017 Oct 19;pii: clincanres.1577.2017.
Epithelial-to-mesenchymal transition antagonizes response to targeted therapies in lung cancer by suppressing BIM
Dacomitinib purchased from AbMole
| Molecular Weight | 469.94 |
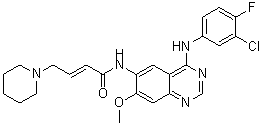
| Formula | C24H25ClFN5O2 |
| CAS Number | 1110813-31-4 |
| Solubility (25°C) | DMSO ≥ 25 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related EGFR/HER2 Products |
|---|
| BBT-207
BBT-207 is a reversible, mutant-specific EGFR inhibitor with antitumor activity. |
| VRN-11
VRN-11 is an EGFR C797S inhibitor. |
| TRX-221
TRX-221 is an EGFR C797S inhibitor. |
| TAS-3351
TAS-3351 is an EGFR C797S inhibitor. |
| Larotinib mesylate hydrate
Larotinib mesylate hydrate is a potent, broad-spectrum, orally active tyrosine kinase inhibitor (TKI) that primarily targets EGFR with an IC50 of 0.6 nM. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
