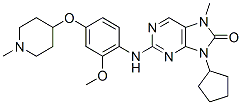
AZ3146 is a novel Mps1 inhibitor with IC50 of 35 nM. AZ3146 significantly inhibits phosphorylation of Mps1 in cells. AZ3146 displays selectivity over 46 other kinases including Cdk1 and aurora kinase B. AZ3146 also inhibits the recruitment of Mad1, Mad2 and centromere protein E (CENP-E) to kinetochores. AZ3146 has a dramatic effect on kinetochore localization of Mad2, reducing its levels to ~15%, but its effect on Mad1 is less pronounced, with levels remaining at ~60%.
| Molecular Weight | 452.55 |
| Formula | C24H32N6O3 |
| CAS Number | 1124329-14-1 |
| Solubility (25°C) | DMSO 25 mg/mL Ethanol 90 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related Kinesin Products |
|---|
| Eg5-IN-1
Eg5-IN-1 is a potent kinesin family motor protein (Eg5) inhibitor with an IC50 value of 1.97 µM. |
| PVZB-1194
PVZB-1194 is a biphenyl-type inhibitor of kinesin Eg5 that induces apoptosis by inhibiting Eg5 ATPase activity and shows potent kinesin spindle protein (KSP) inhibition only in the presence of microtubules, with anticancer activity. |
| Litronesib
Litronesib (LY2523355) is a selective mitosis-specific kinesin Eg5 inhibitor, with antitumor activity. |
| MK-0731
MK-0731 is a selective, non-competitive and allosteric kinesin spindle protein (KSP) inhibitor with an IC50 of 2.2 nM and a pKa of 7.6. MK-0731 is >20,000 fold selectivity against other kinesins. MK-0731 induces mitotic arrest and induces apoptosis in tumors. MK-0731 provides significant antitumor efficacy. |
| BRD9876
BRD9876 is the “rigor” inhibitor that locks kinesin-5 (Eg5) in a state with enhanced microtubules (MTs) binding, leading to bundling and stabilization of MTs. BRD9876 interacts with the tyrosine 104 residue that is part of the α4-α6 allosteric binding pocket. BRD9876 specifically targets microtubule-bound Eg5 and selectively inhibits myeloma over CD34 cells. BRD9876 has the potential for multiple myeloma (MM) research. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
