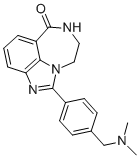
AG14361 is a novel poly (ADP-ribose) polymerase-1 inhibitor with a GI50 of 10.9 µM. AG14361 enhances temozolomide activity in all MMR-proficient cells (1.5–3.3-fold) but is more effective in MMR-deficient cells (3.7–5.2-fold potentiation), overcoming temozolomide resistance. AG14361 significantly potentiated camptothecin-mediated cytotoxicity in all cells, except the base excision repair–deficient EM9 cells. In the breast cancer cell lines, AG14361 had no effect on IR-induced degradation of IkappaBalpha or nuclear translocation of p50 or p65. However, AG14361 inhibited IR-induced NF-kappaB-dependent transcription of a luciferase reporter gene.
| Cell Experiment | |
|---|---|
| Cell lines | LoVo, A549, and SW620 cells |
| Preparation method | Cell growth inhibition was estimated in exponentially growing LoVo, A549, and SW620 cells in 96-well plates. Cells were exposed to temozolomide (0–1000 μM, see Fig. 4, A) or topotecan (0–30 nM, see Fig. 4, B) in the presence or absence of 0.4 μM AG14361. Cells were also exposed to AG14361 (0–20 μM, see Fig. 4, C) alone or in the presence of 400 μM temozolomide. After 5 days of culture, these cells were fixed with 10% trichloroacetic acid and stained with sulforhodamine B (28). The concentration of temozolomide, topotecan, and AG14361 alone or in combination that inhibited growth by 50% (GI50) was calculated from computer-generated curves. |
| Concentrations | 0–20 μM |
| Incubation time | 5 days |
| Animal Experiment | |
|---|---|
| Animal models | CD-1 nude mice bearing palpable, subcutaneous SW620 or LoVo xenografts |
| Formulation | saline |
| Dosages | 5 or 15 mg/kg daily for 5 days |
| Administration | intraperitoneally |
| Molecular Weight | 320.39 |
| Formula | C19H20N4O |
| CAS Number | 328543-09-5 |
| Solubility (25°C) | DMSO ≥12 mg/mL |
| Storage |
Powder -20°C 3 years ; 4°C 2 years In solvent -80°C 6 months ; -20°C 1 month |
| Species | Mouse | Rat | Rabbit | Guinea pig | Hamster | Dog |
| Weight (kg) | 0.02 | 0.15 | 1.8 | 0.4 | 0.08 | 10 |
| Body Surface Area (m2) | 0.007 | 0.025 | 0.15 | 0.05 | 0.02 | 0.5 |
| Km factor | 3 | 6 | 12 | 8 | 5 | 20 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of Compound A used for a mouse (20 mg/kg) to a dose based on the BSA for a rat, multiply 20 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for Compound A of 10 mg/kg.
| Related PARP Products |
|---|
| DSB-1522
DSB-1522 is a PARP1 inhibitor that can be used in tumor-related studies. |
| DM-5167
DM-5167 is a PARP1 inhibitor that can be used in studies related to triple negative breast cancer. |
| NMS-03305293
NMS-03305293 is a PARP inhibitor with high selectivity for PARP1 isoforms and low DNA capture effect, which specifically kills BRCA mutated tumor cells. In addition, NMS-03305293 can penetrate the blood-brain barrier and can be used in studies related to CNS tumors and brain metastatic tumors. |
| SNV1521
SNV1521 is a potential best-in-class (BIC), highly selective and CNS-permeable PARP1 inhibitor. |
| Basroparib
Basroparib is an orally active selective inhibitor of end-anchor polymerase (TNKS) and an inhibitor of ribose polymerase (PARP) with antitumor activity. |


Products are for research use only. Not for human use. We do not sell to patients.
© Copyright 2010-2023 AbMole BioScience. All Rights Reserved.
